Question
Question: With conc. \({\rm{HBr}}\) ethylphenyl ether yields; A. Phenol and ethyl bromide B. Bromobenzene ...
With conc. HBr ethylphenyl ether yields;
A. Phenol and ethyl bromide
B. Bromobenzene and ethanol
C. Phenol and ethane
D. Bromobenzene and ethane
Solution
Alkyl ethers cleaved by strong acids such as HBrandHI in nucleophilic substitution reaction. The protonation of ethereal oxygen produces a good leaving group along with the neutral alcohol molecule. Depending on the structure of the alkyl group, the reaction can be SN2.
Complete step by step answer:
The ether molecules have net dipole moment due to the polarity C−O bon d.The boiling points of ethers are comparable to alkanes but much lower than alcohols of comparable molecular masses. Ether molecules are miscible in water due to their polar nature (‘‘like dissolves like’’ rule i.e. polar molecule dissolves in polar solvent and nonpolar molecule will be dissolved in nonpolar solvent).
Diethyl ether used as a solvent in organic chemistry. Vapours of some ethers used as miticides, insecticides and fumigants for soil.
Ethyl phenyl ether reacts with conc. HBr to give alkyl halide and alcohol. The mechanism involves two steps as:
Step 1: Protonation of the alcoholic oxygen takes place to make a better leaving group. This step is very reversible and fast.
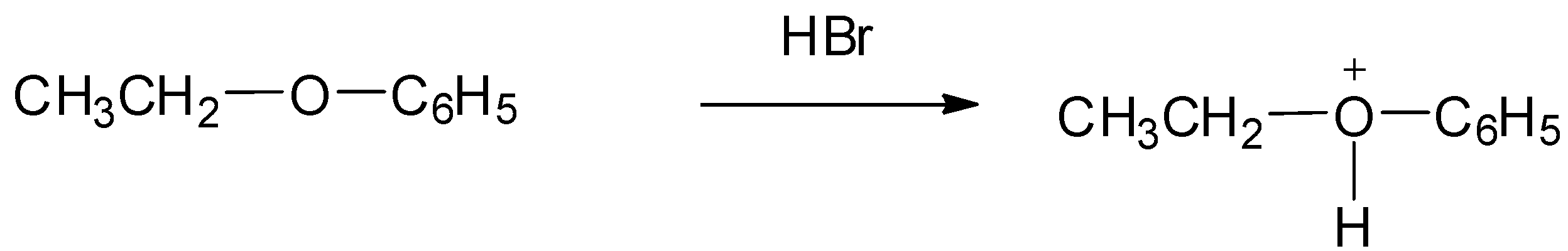
Step 2: The bromide ion acts as nucleophile and attacks to displace the good leaving by cleaving the C−O bond producing neutral alcohol and alkyl bromide.
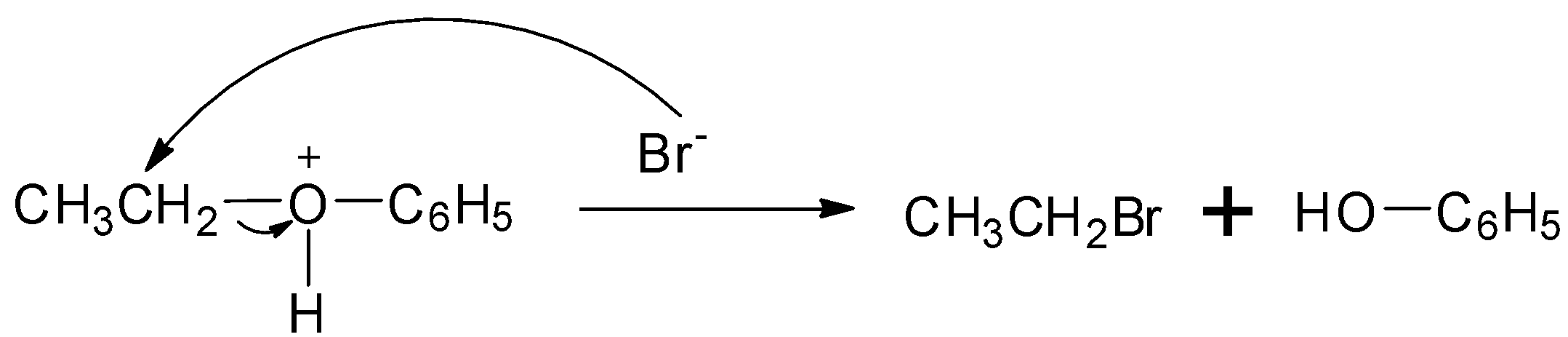
So, the correct answer is Option A.
Note: The leaving group in the above mechanism must be stable. In the above case, the second step is the slowest step and rate determining step. CH3CH2+ carbocation is more stable than C6H5+ because the positive charge in C6H5+ is localized over the ring.
