Question
Question: Williamson’s synthesis uses _ _ _ _ _ _ mechanism. (A)- \({{S}_{N}}1\) (B)- \({{S}_{N}}2\) (...
Williamson’s synthesis uses _ _ _ _ _ _ mechanism.
(A)- SN1
(B)- SN2
(C)- either SN1 or SN2
(D)- none
Solution
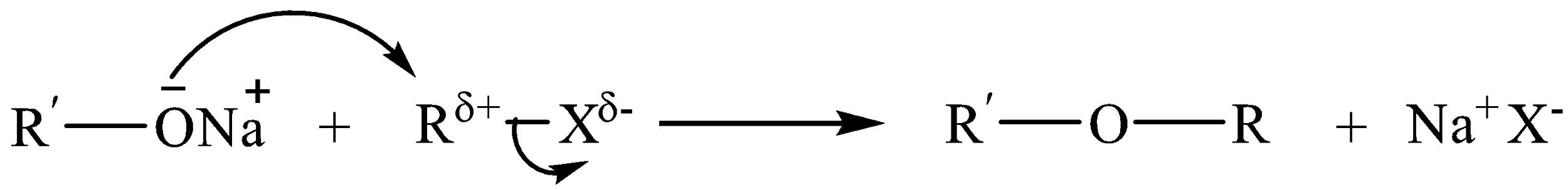
Williamson’s synthesis is one of the best processes used for the synthesis of both symmetrical and unsymmetrical ethers. Alkyl halides react with sodium alkoxide to form ethers. General reaction for the formation of ether by Williamson’s synthesis can be written as
RO−Na−+R′−X→R−O−R′+Na+X−
Complete step by step answer:
Williamson’s ether synthesis involves the preparation of ethers by nucleophilic substitution reaction.
In alkyl halides, carbon halogen bond is polar due to higher electronegativity of the halogen atoms. As a result, positive charge develops on the carbon attached to the halogen atom making it somewhat electrophilic in nature.
Rδ+−Xδ−
Alkoxide ion acts as a nucleophile and substitutes the halide ion from an alkyl halide. The reaction occurs by bimolecular nucleophilic substitution, i.e. SN2 mechanism.
Let us have a look at some of the examples.
Preparation of symmetrical ethers
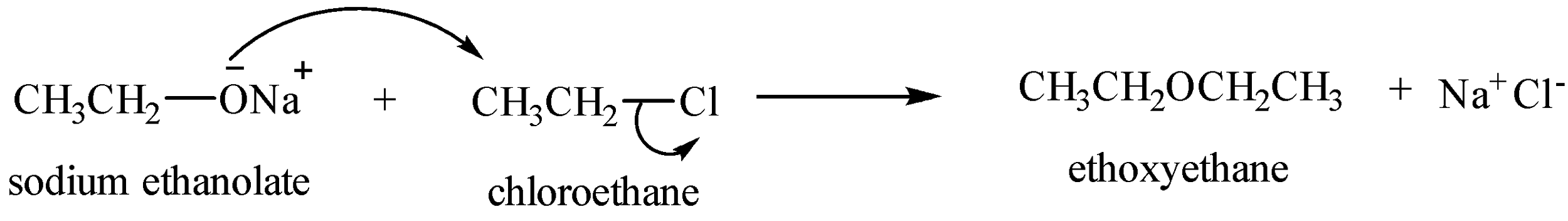
Preparation of unsymmetrical alkenes
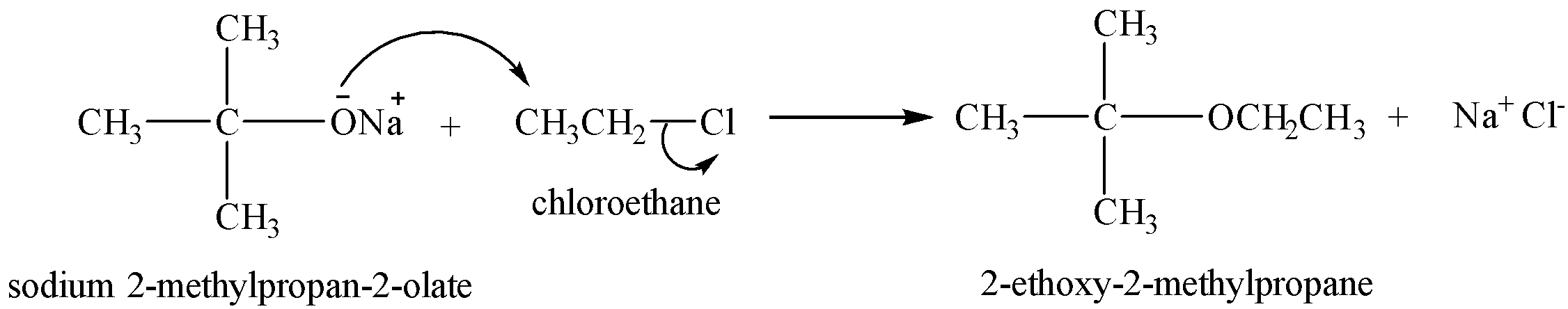
For the preparation of unsymmetrical ethers, primary alkyl halides are taken.
The reaction proceeds via SN2 mechanism, where the nucleophile attacks the carbon from the back side, so if secondary or tertiary alkyl halides are used, the nucleophile attack is hindered by the alkyl groups on the 2o and 3o carbons.
Therefore, in case of secondary and tertiary alkyl halide, the alkoxide ion behaves as a base and abstracts the β-hydrogens, thus resulting in the formation of an alkene.
Since the reaction follows SN2 mechanism, the reactivity of alkyl halides in Williamson’s synthesis follows the same order as that inSN2 mechanism, i.e. 1o>2o>3o
So, the correct answer is “Option B”.
Note: Remember that SN2 reaction is a one step reaction and bond breaking and making takes place simultaneously, hence no intermediate is formed. Due to the backside attack of the nucleophile on alkyl halide the configuration of the product formed is reversed.
