Question
Question: Which of the following compounds is most reactive for Wurtz Reaction? (A) \(C{{H}_{3}}CHBrC{{H}_{3...
Which of the following compounds is most reactive for Wurtz Reaction?
(A) CH3CHBrCH3
(B) 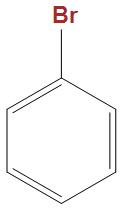
(C) 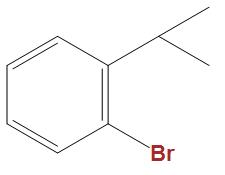
(D) CH3C(CH3)2Br
Solution
As we know that Wurtz reaction is a coupling reaction, which is used to prepare symmetrical alkenes. It is basically a reaction in which higher alkanes are synthesised by a reaction in between alkyl halides and metallic sodium. This reaction takes place in presence of dry ether.
Complete answer:
- As we know that less stable radical is more reactive towards Wurtz reaction. We can see the order of reactivity towards Wurtz reaction as:
primary carbocation (least stable) > secondary carbocation (less stable) > tertiary carbocation (more stable)
- We can see that in CH3CHBrCH3 , there is secondary carbocation present, which is less stable as compared to all other given compounds. Hence, is much reactive towards Wurtz reaction.
- In the option b and c we can see that these are well stabilised by the phenyl ring, hence is not that much reactive towards Wurtz reaction as compared to CH3CHBrCH3.
- In CH3C(CH3)2Br we can see that there is tertiary carbocation present, which is most stable as compared to all other given compounds. Hence, it is not that much reactive towards Wurtz reaction as compared to other given options.
- Hence, we can conclude that the correct option is (a), that is CH3CHBrCH3 most reactive for Wurtz Reaction.
Note: - It is found that methane is not prepared by this reaction. The minimum number of carbon atoms required is two which is not applicable in the case of methane. Hence, this means that ethane is the lowest alkane developed through this reaction.
