Question
Question: Which among the following molecules can exhibit tautomerism? 
A. Both I and II
B. Both II and III
C. III only
D. Both I and III
Solution
Tautomers are structural isomers of each other. They differed from each other in the position of the proton and electron. Ketone functional group having α hydrogen undergoes keto-enol tautomerism. All the given molecules are bridge compounds so use Bredt’s rule and determine which molecule undergoes tautomerism.
Complete answer:
Keto-enol tautomerism is the conversion of a ketone into enol form. There is a chemical equilibrium between both forms.
First, we will write the possible enol isomers for all three molecules.
To convert keto form to enol form compound should contain α hydrogen. So we will assign the α hydrogen to all three structures.
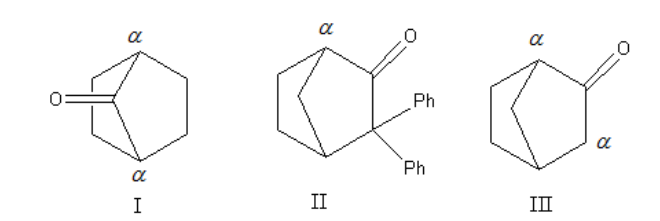
Now, we will write the tautomerism reaction for all three compounds and will predict the possible products.
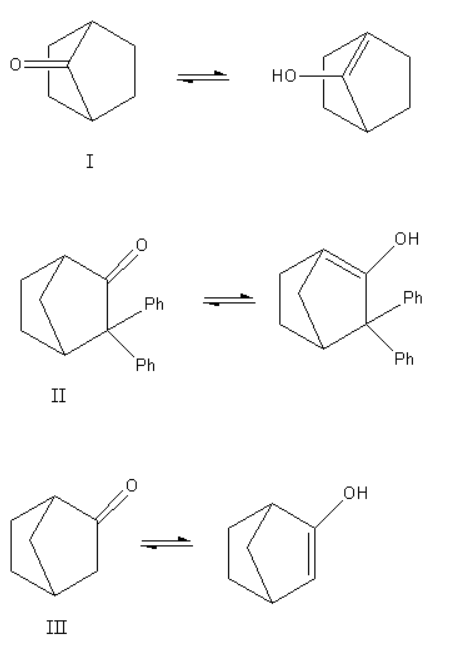
Here, we can see that structure I and II give the bridgehead double bond. According to Bredt’s rule in the case of a bridged ring, the bridgehead double bond does not exist as the bridgehead double bond is unstable. As the enols of structure, I and II have bridgehead double bonds they do not exist. Only structure III gives a non-bridgehead double bond so it shows keto-enol tautomerism.
Thus, out of the three structures only structure III exhibit tautomerism.
**So, the correct option is (C) III only.
Note:**
The molecule should obey two conditions to undergo tautomerism. The first condition is that the molecule should have α hydrogen. The second condition is for bridge ring molecules. If the molecule is a bridged ring the double bond should not be bridgehead.
