Question
Question: What is the colour of methyl orange in acidic and basic medium respectively? (A) Yellow, Red (B)...
What is the colour of methyl orange in acidic and basic medium respectively?
(A) Yellow, Red
(B) Red, Yellow
(C) Colourless, Red
(D) Colourless, Yellow
Solution
Methyl orange is an organic compound which is used as a laboratory agent. It is mostly used as a pH indicator frequently used in titrations due to its distinct colour and variance at different pH values.
Complete step by step answer: Methyl orange is a chemical compound having chemical formula C14H14N3NaO3S.
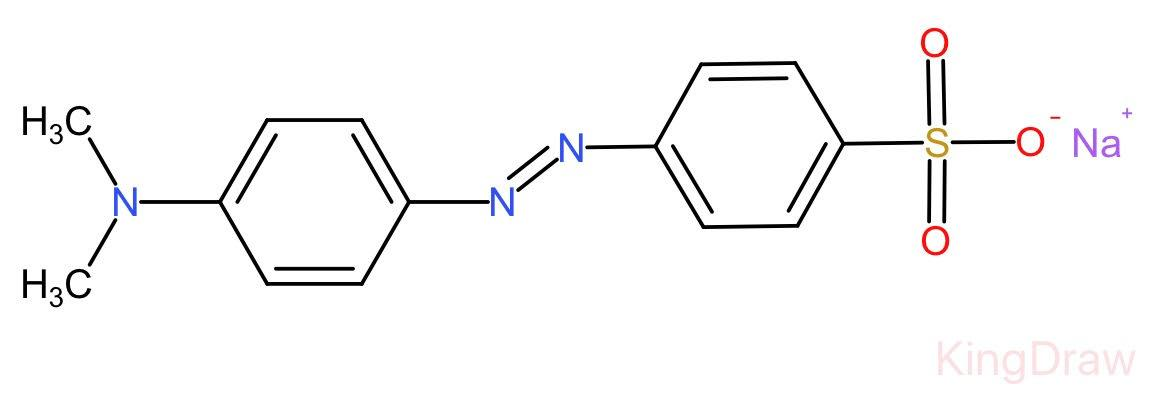
Unlike a universal indicator, it does not show a full spectrum of colour change but does have a sharp endpoint.
- In a solution that is becoming less acidic in nature, methyl orange tends to change its colour from red to orange and finally to yellow. This entire colour change occurs in acidic conditions.
- Methyl orange shows a red colour when the pH of the solution is less than or equal to 3.1 and shows a yellow colour when the pH is above or equal to 4.4.
- As the changes occur in the pka of a mid-strength acid, methyl orange is usually used for the titration of acids.
Therefore, in the acidic medium, it is red in colour and in the basic medium, it is yellow in colour.
Hence, option (b) is the correct answer.
Note: Methyl orange is insoluble in diethyl ether, but its solubility in water is 0.5 g/100mL (20 degree Celsius). It is an orange solid in appearance and direct contact with methyl orange should be avoided.
