Question
Question: What is Compound A? 
A) CH3CH2CH2CH=CH2
B) CH3CH2CH(CH3)CH=CH2
C) CH3CH2CH=CHCH3
D) CH3CH2C(CH3)=CHCH3
Solution
The given reaction is the addition of HBr in an alkene. The addition should take place in presence of an alkene. Hence compound A should be an alkene.
Complete answer: Markovnikov's rule helps us predict the regioselectivity of the addition of an electrophile in alkenes and alkynes. This rule states that, in Hydrohalogenation of an unsymmetrical alkene, the negative part of the substrate will get attached to that carbon atom having less no. of hydrogen and the positive part of the substrate gets attached to that carbon having more no. of hydrogen.
The product given to us is 3-bromo 3-methyl pentane. To obtain this product via addition of HBr, the reactant should have a double bond between C2&C3. Such a case is fulfilled in Option D).
The addition of HBr proceeds via an ionic mechanism and forms a carbocation intermediate. The H+ from HBr will attack on carbon two i.e. C2 and form a tertiary carbocation on C3 . The Br of the HBr will then attack on the tertiary carbocation to form the desired product. The reaction can be given as:
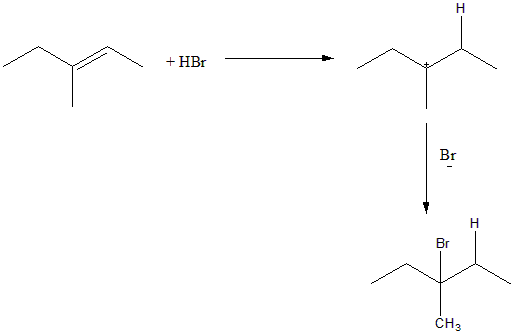
Hence the correct answer is Option (D).
Note:
If peroxide was added in the reaction, then we would have got 2-bromo,3-methyl pentane which would be an Anti Markovnikov Product. This happens only in the presence of peroxide. The reaction that would occur is a free radical addition reaction. Peroxides from free radicals that initiate the reaction. A strong source of light is required to break the -O-O- peroxide bond, like sunlight, floodlight or UV light.
