Question
Question: What is A in the following reaction? 
Solution
Remember to apply Markovnikov’s rule whenever a protic acid is added to an asymmetric alkene. The rule states that: In addition reactions involving unsymmetrical alkenes, the electron rich component of the reagent adds to the carbon atom with less number of hydrogen atoms bonded to it. The electron deficient component adds to the carbon with more hydrogen atoms bonded to it.
Complete answer:
The given reaction proceeds with Markovnikov’s rule. First, electrophilic addition takes place and then nucleophilic attack occurs. According to Markovnikov’s rule, the electron rich component of the reagent adds to the carbon atom with less number of hydrogen atoms bonded to it. Nucleophilic part is the electron rich component of a reagent. Nucleophiles are chemical species which form bonds with electrophiles by donating an electron pair. All molecules and ions which have a free pair of electrons or at least one pi bond can act as nucleophiles. The nucleophile attaches to the carbon having less number of hydrogen atoms. Cl− is the nucleophile in the given reagent. The middle carbon has less number of hydrogens attached to it. So, the nucleophile attaches to the middle carbon. The reaction is given below:
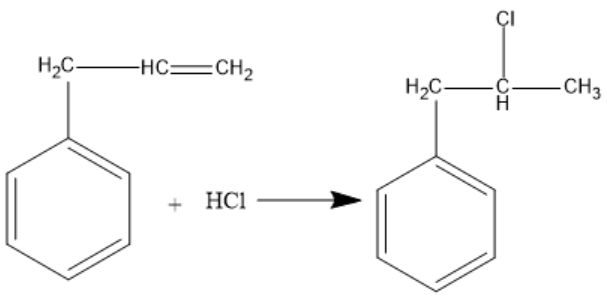
Note:
While solving questions which involve addition of protic acids to unsymmetrical alkenes, we have to apply Markovnikov's rule and identify the nucleophilic part in the given reagent. Then, look for the carbon in the unsymmetrical alkene which has less number of hydrogens attached to it and attach the nucleophile to it.
