Question
Question: What are the products of hydrolysis of sucrose?...
What are the products of hydrolysis of sucrose?
Solution
Sucrose is the common table sugar we use in our daily lives. It is a disaccharide, i.e. it is composed of two sugars by a glycosidic linkage. The monosaccharides involved in the structure of sucrose are both six carbon compounds.
Complete answer:
Sucrose is a saccharose, also known by names such as table sugar, cane sugar, beet sugar, etc.
It is a carbohydrate, more specifically, a disaccharide, made up of two monosaccharides, namely, glucose and fructose.
Glucose is an aldehyde, which forms a six-carbon ring.
Whereas, fructose is a ketone, which forms a five-membered ring (hence, a furan).
Therefore, on hydrolysis sucrose will break into its monomeric sugars. The reaction can be represented as –
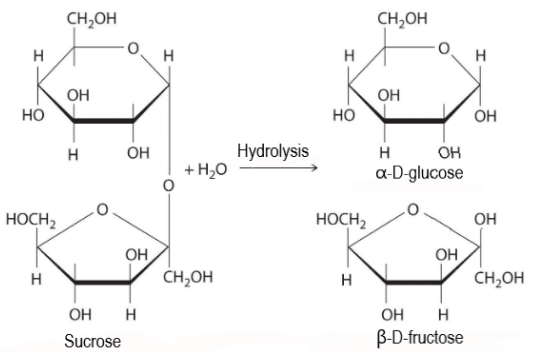
As we can see, sucrose is made by linking C1 of alpha-glucose and C2 of beta-fructose. The bond formed between glucose and fructose is known as 1,2-glycosidic bond.
Also, sucrose is a dextrorotatory sugar, which changes to levorotatory, due to the dominant levorotatory nature of fructose.
Therefore, the answer is – the products of hydrolysis of sucrose are glucose and fructose.
Additional Information: Fructose is the sweetest natural sugar.
Note: Disaccharides are reducing sugars, except sucrose. Sucrose is not a reducing sugar because the reducing groups of glucose and fructose are involved in glycosidic bonds. It, therefore, gives a negative Tollens test.
