Question
Question: What are the isomers in relation to glucose, galactose and fructose?...
What are the isomers in relation to glucose, galactose and fructose?
Solution
Carbohydrates are sugars containing the atoms of oxygen, hydrogen and oxygen. The general molecular formula of carbohydrates is Cx(H2O)y . The hydrogen atoms will be double the oxygen atoms. Glucose, galactose and fructose are carbohydrates and differ in structure, called structural isomers.
Complete answer:
Glucose is a carbohydrate with the molecular formula of C6H12O6 . Galactose is also having a same molecular formula of C6H12O6 , fructose has also same molecular formula C6H12O6 . But the cyclic structure of glucose, galactose and fructose differ in structures. Hence, these three are called structural isomers.
The glucose is having a hydroxyl group and aldehyde group. The fructose is having a hydroxyl group and ketone group. The galactose has the same configuration as glucose, but there is a difference of hydroxyl group attached direction at C4 carbon.
The cyclic structure of glucose, galactose and fructose are:
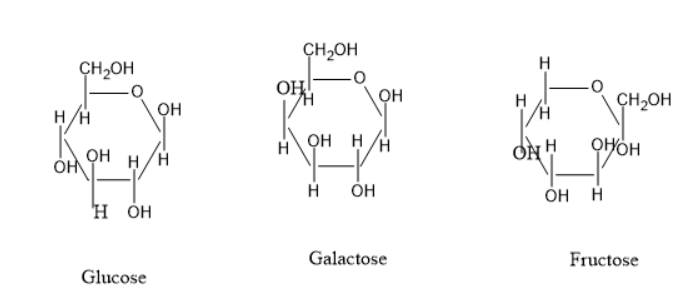
The above three structures have the same molecular formula but there is a difference at the structures, hence these are called structural isomers.
Glucose and galactose have the difference of hydroxyl group position at C4 carbon. The fructose and glucose also have a difference in structure. Thus, these all are structural isomers.
Note:
The glucose and fructose have differences in functional groups in normal structure. But, in cyclic structures there is no difference in functional groups but differ only in structure and can be called structural isomers. The galactose glucose is also having different hydroxyl group positions.
