Question
Question: What are delocalized electrons?...
What are delocalized electrons?
Solution
The phenomena of hyperconjugation and resonance are the property of compounds having alternate double and single bonds. These alternate double and single bonds consist of delocalization of electrons. Delocalization means the movement of electrons to acquire stable positions in the same molecule.
Complete answer:
The movement of electrons that are not in a bonded state inside a molecule that results in a highly stable molecule is the phenomena of delocalization of electrons. The electrons involved in the delocalization are called delocalized electrons.
Delocalization occurs when the pi bonds provide extra energy, so that the non – bonded electrons move in the whole molecule. The delocalized electrons do not remain in one place and tend to rotate; this provides the molecule with extra stability. A diagrammatic representation of the delocalization of electrons in benzene is,
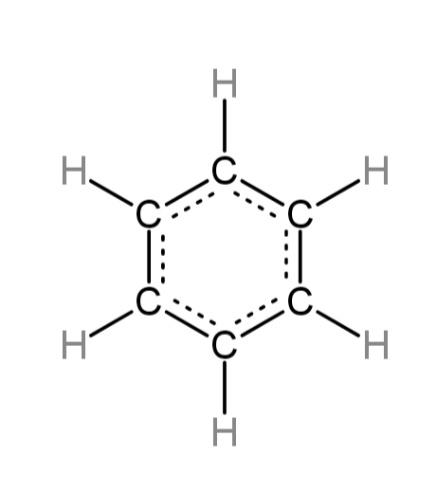
The double bonds contain the pi bonds that are made of electrons which are loosely held, this creates the loosely held electrons to move and hence they become delocalized. The delocalization results in the higher stabilization of energy in the molecule, for example in benzene molecule as follows:
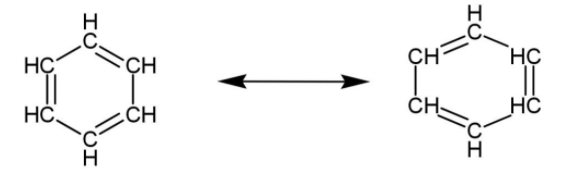
Due to delocalization of electrons, the two resonating structures of benzene are formed. As a result the bond length of the bonds in benzene are the same, that gives this molecule extra stability.
Hence, delocalized electrons are the electrons that are free and move inside a molecule to provide it with extra stability.
Note:
Since delocalization involves the pi electrons, hence delocalization can be seen in compounds having alternate double and single bonds. The more the delocalization, more the stability of the compound. The delocalization results in resonance, which increases the stability and lowers the energy of the compound.
