Question
Question: The synthesis of 3-octyne is achieved by adding a bromoalkane into a mixture of sodium amide and an ...
The synthesis of 3-octyne is achieved by adding a bromoalkane into a mixture of sodium amide and an alkyne. The bromoalkane and alkyne respectively are:
(a) BrCH2CH2CH2CH3andCH3CH2C≡CH
(b) BrCH2CH2CH3andCH3CH2CH2C≡CH
(c) BrCH2CH2CH2CH2CH3andCH3C≡CH
(d) BrCH2CH2CH2CH3andCH3CH2C≡CH
Solution
By analyzing the given options we know that the final product that will be formed will be a terminal alkane and an alkylhalide.3-octyne has eight carbon atoms in their structure in which the third carbon will have a triple bond.
Complete step-by-step answer: Now let us find the data provided in the question. The question is all about the bromoalkane and the alkyne that has been involved in the synthesis of 3-octyne.To get the appropriate bromoalkane and alkyne that were involved in the synthesis, we have to cleave the molecule. And we know that sodium amide is involved, so the synthesis of 3-octyne using the alkyne and bromoalkane was a SN2reaction. And as the sodium amide is involved the alkyne involved must be terminal, then only it could abstract the acidic hydrogen.
Now let us discuss the structure of 3-octyne, which will give us an idea of the reagents. So 3-octyne is a molecule that will have eight carbon atoms in their parent chain since the prefix –oct represents 8. And as in the name it is given that 3-octyne, '3' represents the position of the triple bond. The suffix –one represents a triple bond. So in the molecule triple bond will be present in the third carbon.
The structure of 3-octyne is as follows:
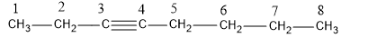
Now we could cleave the molecule either from the third carbon (C-3) or from the fourth carbon (C-4).
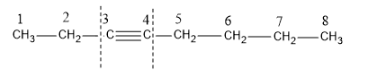
Now let us see the products formed if cleavage takes place at C-3.
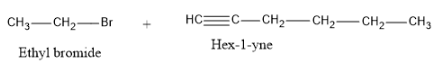
And now let's see the products formed if the cleavage takes place at C-4.
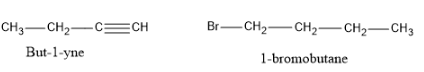
If we compare these answers with the options given, the correct set of options from the given options is option (d).
Note: One should be thorough with the nomenclature of the compounds since we have to draw the structure and should have a clear idea of the numbering of each atom. And one should practice the mechanism involved in the cleavage of a molecule and how it will undergo cleavage at different conditions and with different reagents.
