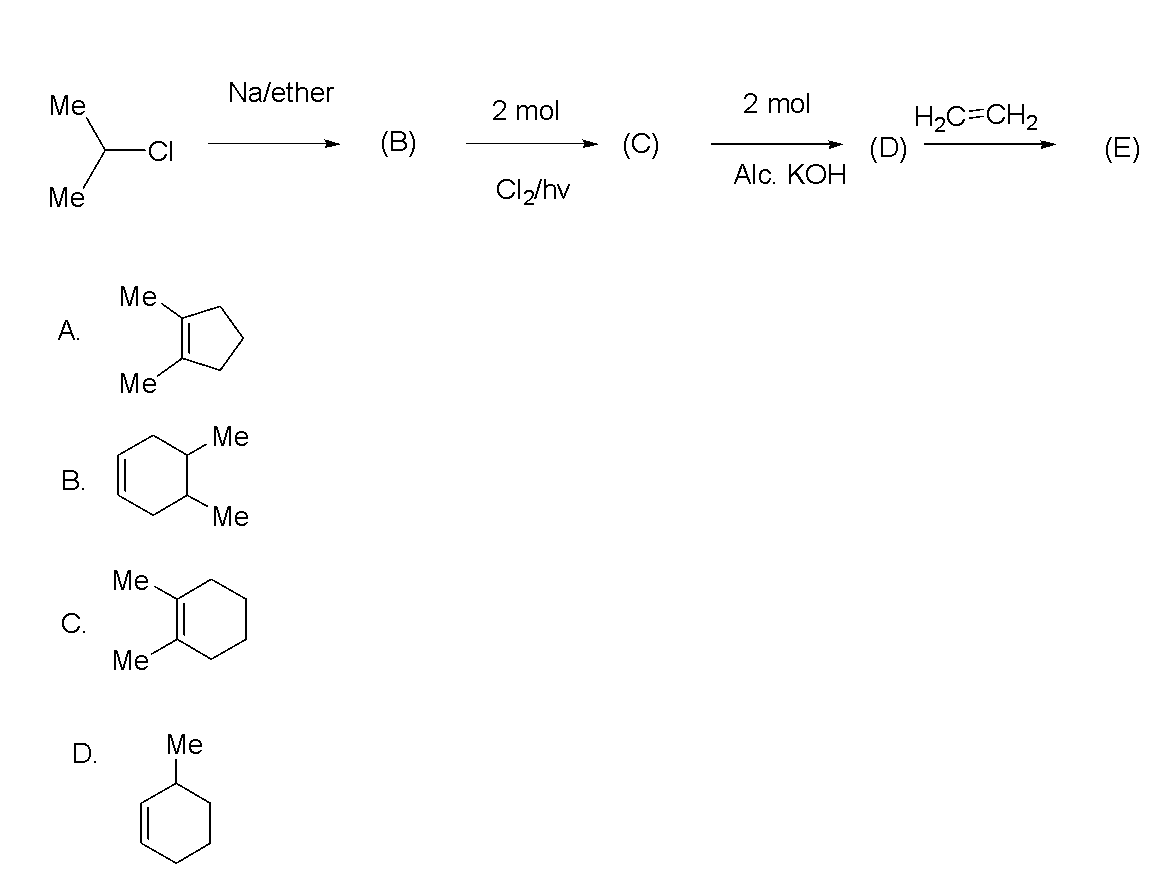
Question
Question: The final product (E) is  is
Solution
Hint : We must understand that in the first step of the reaction, a haloalkane treated with Na/ether is a typical Wurtz reaction, therefore the product B is dimer of the alkane part of the haloalkane. In the second step, the alkane obtained in the previous step is treated with 2 moles of Cl2/ light, here free radical substitution reaction occurs. The product obtained in this step is dihaloalkane (C). In the third step, C is treated with 2 moles of alcoholic KOH, this is a typical dehydrogenation reaction, i.e. removal of two moles of HCl occurs. As a result of this, we obtain a diene which undergoes Diels Alder cycloaddition reaction.
Complete step by step solution :
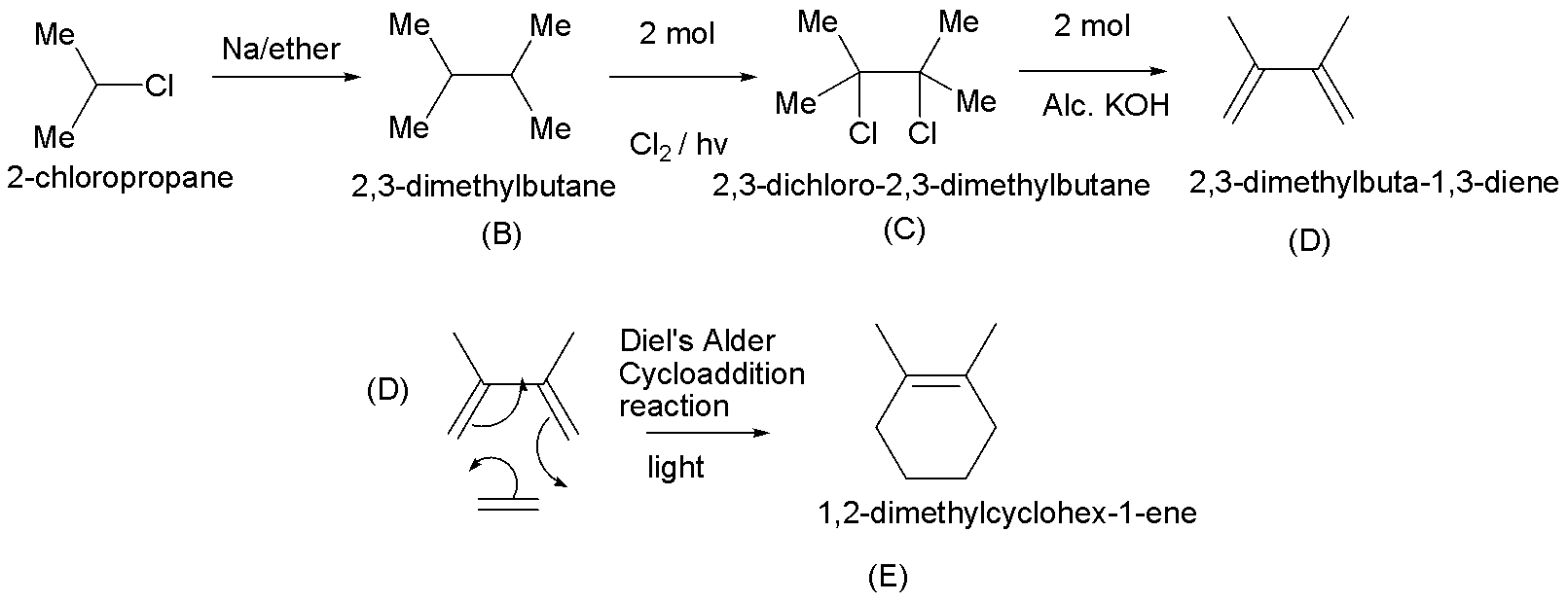
Step 1:- In this step, a haloalkane that is treated with Na/ether is a typical Wurtz coupling reaction. It is one of the oldest organic reactions, and produces the simple dimer derived from two equivalents of alkyl halide. Therefore the product obtained is 2,3-dimethylbutane (B).
Step 2:- Here, 2,3-dimethylbutane (B) is treated with 2 moles of chlorine in the presence of light. This is a free radical substitution reaction. The product obtained is 2,3-dichloro-2,3-dimethylbutane (C).
Step 3:- In this step, 2,3-dichloro-2,3-dimethylbutane undergoes dehydrohalogenation in the presence of strong bases such as alcohol KOH. Hence, the product in this reaction is 2,3-dimethylbuta-1,3-diene (D).
Step 4:- The obtained diene (D) undergoes Diels Alder cycloaddition reaction in the presence of light, hence the product (E) is 1,2-dimethylcyclohex-1-ene.
Hence, the correct option is C.
Note : 1. In Wurtz reaction, dry ether is used because sodium metal is used which is highly reactive with water. Therefore, we use dry solvent which does not react with sodium metal. Thus, dry ether is used.
2. In step 3, here dehydrohalogenation takes place as a type of E2elimination reaction favoured in the presence of a strong base.
3. Step 4, cycloaddition reaction is the concerted bond formation between two independent pi-electron systems to form a new ring of atoms. Here, the Diels-Alder cycloaddition is classified as a [4+2] process because the diene has four pi-electrons that shift position in the reaction and the dienophile has two.
