Question
Question: The correct structure of monomers of buna-N is : (A)  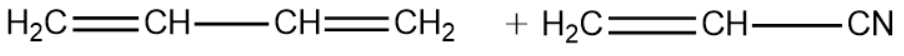
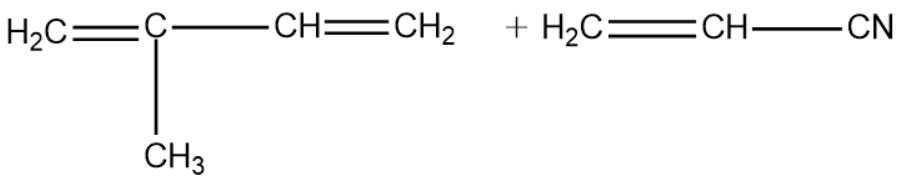
(B)
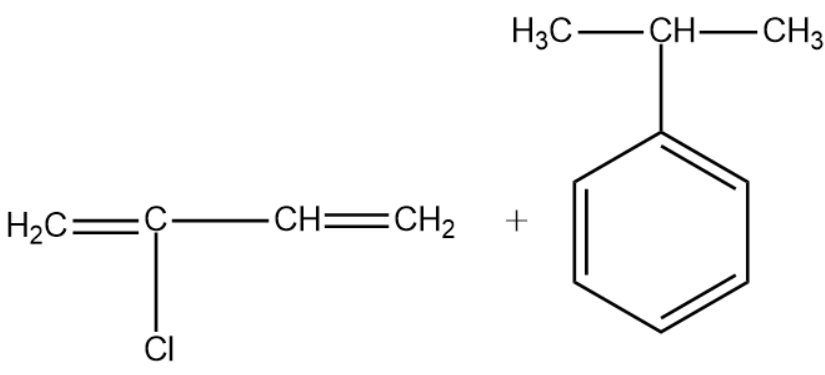
(C)
(D) 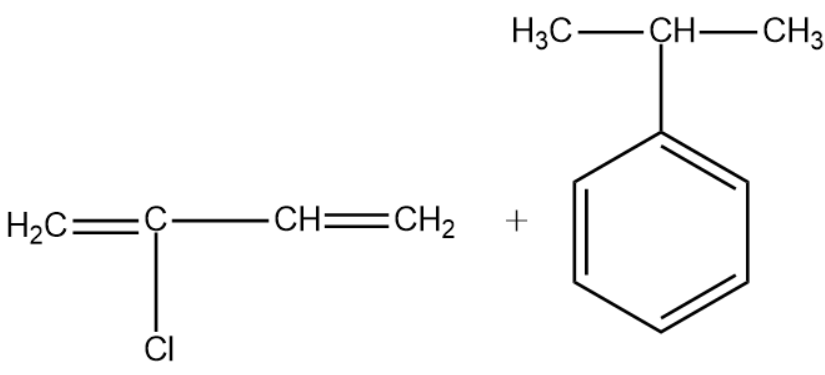
Solution
Buna-N is a polymer made up of 2 polymers. This means that it is a copolymer. The reaction mechanism involves formation of free radicals. So, check for compounds which can easily generate free radicals. Identify the names of the monomers and then draw the structures accordingly.
Complete step-by-step answer:
Polymerization is a process of reacting monomer molecules together in a chemical reaction to form long polymer chains .
In chemical compounds, polymerization can occur by various types of reaction mechanisms that vary in complexity due to the functional groups present in the reactants and the steric effects that are present.
Copolymerisation is the type of polymerisation reaction involving more than one type of monomer. The number of monomers involved in the reaction is usually 2.
Buna-N is one such polymer that is formed by copolymerisation. The monomers of the polymer are 1,3-Butadiene and vinyl cyanide.
We will now draw the structures of each monomer and then identify the correct answer.
1,3-Butadiene:
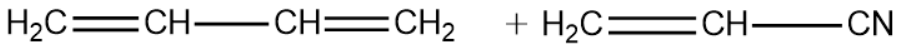
Vinyl Cyanide:
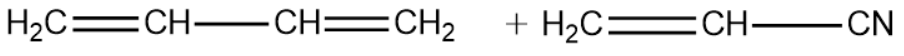
We will now write the polymerisation reaction of the above two monomeric units and see the formation of the Buna-N polymer.
From the above explanation we can conclude that the correct answer is option (A).
Note: In simple polymerizations, alkenes form polymers through relatively simple free radical reactions; in contrast, reactions involving substitution at a carbonyl group require more complex synthesis due to the way in which reactants polymerize and the steric hindrance offered by neighboring groups.
