Question
Question: Tautomerism is exhibited by: This question has multiple correct options. (A) \( {(M{e_3}CCO)_3}C...
Tautomerism is exhibited by:
This question has multiple correct options.
(A) (Me3CCO)3CH
(B) 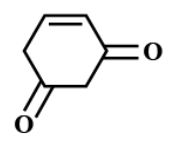
(C) 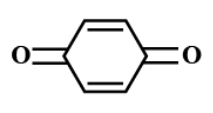
(D)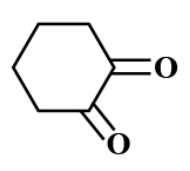
Solution
Tautomers are isomers of a compound which differ only in the position of the protons and electrons. Molecules that exhibit tautomerism have the same molecular formula and they interconvert rapidly.
Complete answer:
Tautomerism is exhibited by those compounds which have α− hydrogen atoms. The α− carbon in an organic molecule refers to the first carbon atom that attaches to a functional group, such as a carbonyl group. A hydrogen atom attached to an α− carbon atom is called an alpha-hydrogen atom. In tautomerism, there is an exchange of a hydrogen atom between two other atoms while forming a covalent bond to either one.
Now, if we look in the above given options, we can see that in option A,
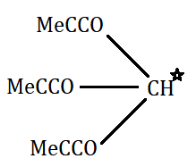
there is one α− hydrogen atom in (Me3CCO)3CH . While in option B and option D, there are two α− hydrogen atoms.
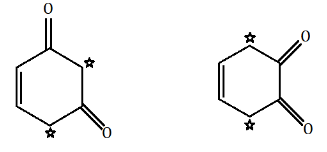
There is no α− hydrogen atom present in option C.
Hence, tautomerism is exhibited by compounds of option A, option B and option D.
The α− hydrogen atoms are represented using star marks and they enable the compounds to show keto-enol tautomerism.
To exhibit tautomerism, a compound must contain polar molecules and weak acidic functional groups. Tautomerism provides more stability to a compound. Tautomerism is a reversible process. Some examples of tautomerism are keto-enol, enamine-imine, lactam-lactim configurations etc.
Note:
Tautomerism involves proton transfer in an intramolecular fashion. Tautomerism has no effects on bond length or any such features. Tautomerism generally occurs in planar or non-planar molecules. Students should be careful not to confuse tautomers with depictions of "contributing structures" in chemical resonance. Resonance structures are merely convenient depictions and do not physically exist.
