Question
Question: Reaction of \({\text{RCON}}{{\text{H}}_{\text{2}}}\) with a mixture of \({\text{B}}{{\text{r}}_{\tex...
Reaction of RCONH2 with a mixture of Br2 and KOH gives RNH2 as the main product. The intermediate involved in the reaction is:
(A)
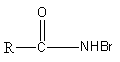
B.R−NHBr
(C)
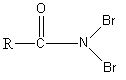
Solution
: The amide can be converted into amine by reacting the amide with bromine and sodium hydroxide. The reaction of conversion of primary amide into amine in presence of bromine with potassium hydroxide is known as Hoffmann bromamide reaction. To determine the answer we should know the mechanism of the reaction.
Complete step by step solution: Hoffmann bromamide reaction is used for the preparation of primary amine from amide. The reaction is shown as follows:
RCONH2 + Br + KOH→RNH2 + 2NaBr + K2CO3 + 2H2O
The mechanism of the reaction is as follows:
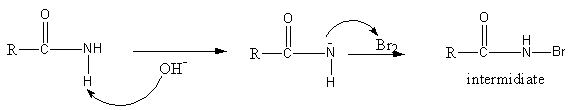
Initially, the base sodium hydroxide abstract proton from amide to generate an anion of amide. The anion of amide now attacks on bromine, so an N-bromoamide generates.
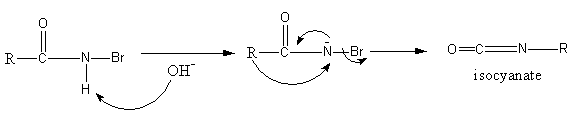
Again the base attacks on N-bromoamide so an anion of bromoamide forms. Then the methyl group attached with the carbonyl group shifts to the nitrogen atom and bromide ion leaves forming an isocyanate structure.
Nucleophilic attack of water on isocyanate takes place which loses the carbon dioxide and forms a structure in which nitrogen has a negative charge and one hydrogen and one methyl group. Then the protonation of this nitrogen takes place which gives amine.
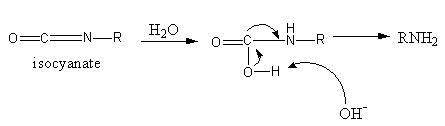
So, the intermediate involved in the reaction is,
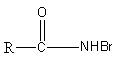
Therefore, option (A) is correct.
Note: The Hoffmann bromamide reaction is used for the preparation of primary amine only. One important structure form during the reaction is isocyanate. Lossen rearrangement, Beckmann rearrangement and Hofmann reaction, all are used for the preparation of primary amine. Hoffmann bromamide reaction and Lossen rearrangement are used for the preparation of primary amine only via the formation of isocyanate. Beckmann rearrangement gives amides from oximes.
