Question
Question: Phenylacetylene on treatment with \(HgS{{O}_{4}}/{{H}_{2}}S{{O}_{4}}.{{H}_{2}}O\) produces: A. Ace...
Phenylacetylene on treatment with HgSO4/H2SO4.H2O produces:
A. Acetophenone
B. Phenylacetaldehyde
C. Phenylacetic acid
D. 1-Phenylethanol
E. 2-Phenylethanol
Solution
Generally alkenes reacts with strong acids and forms respective alcohols or ketones as the product. But alkynes won’t react with strong acids, alkynes undergoes hydration reaction in the presence of mercuric sulphate catalyst and forms the respective ketone or aldehyde as the product.
Complete answer:
- In the question it is given that the treatment of phenylacetylene with HgSO4/H2SO4.H2O produces which product.
- Reaction of alkynes with HgSO4/H2SO4.H2O is an example for hydration reaction on alkynes using mercuric sulphate as the reagent.
- The reaction of phenylacetylene with HgSO4/H2SO4.H2O is as follows.
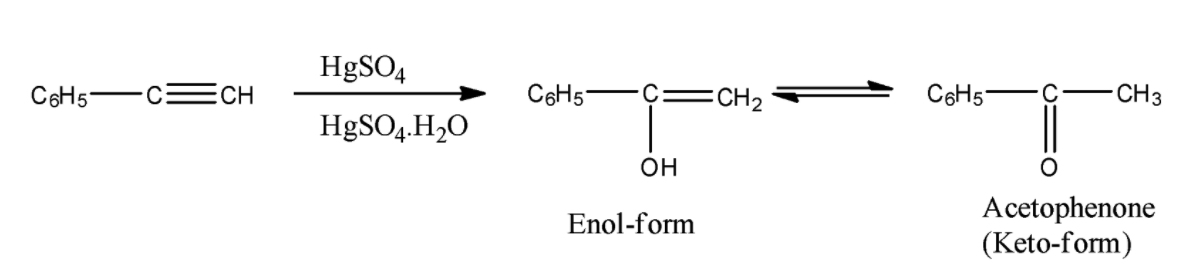
- In the above reaction phenylacetylene reacts with HgSO4/H2SO4.H2O and forms an enol as the product.
- We know that enol form is unstable at room temperature and converts to keto-form and the product name is acetophenone.
- Therefore the reaction of Phenylacetylene with HgSO4/H2SO4.H2O produces acetophenone as the product.
- So, the correct option is A.
Additional information:
- The reaction of alkynes with HgSO4/H2SO4.H2O and produces a aldehyde or ketone as the product is called Kucherov’s reaction.
- This is a type of hydration reaction and it is not possible by using strong acids as the reagent.
- Strong acids can react with alkenes and form respective products.
Note: If the alkynes are highly substituted then the alkynes gives ketone as the product on reaction with HgSO4/H2SO4.H2O. If the alkynes are less substituted then the less substituted alkynes gives aldehyde as the product.
