Question
Question: Identify most acidic hydrogen in given compound. 
(A)- c
(B)- b
(C)- a
(D)- d
Solution
The acidic hydrogen means the hydrogen atom which will easily release from the compound and make the solution acidic. So, the hydrogen atom which will form a stable resonating structure after its removal will be the most acidic hydrogen atom.
Complete step-by-step answer: We know that the solution becomes acidic when there is the presence of hydrogen ions (H+) in the solution. The acidic hydrogen means the hydrogen atom which will easily release from the compound and make the solution acidic. The given compound the question is:
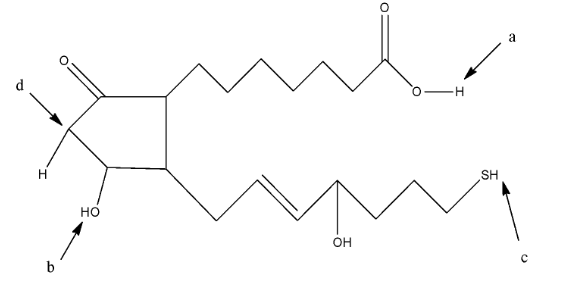
In this compound, we have to find the most acidic hydrogen. So, the hydrogen atom which will form a stable resonating structure after its removal will be the most acidic hydrogen atom. This is due to the fact that the resonance in the compound will make the compound stable.
The hydrogen atom attached to the acidic group will be the most acidic because the hydrogen atom is attached with very electronegative atom oxygen. In a, the hydrogen atom is attached with an oxygen atom which is further attached with a carbon atom having a double bond, so it is an acidic hydrogen. In b, the hydrogen atom is attached to the oxygen atom but it is not further attached to a carbon atom. In c, the hydrogen atom is attached to a sulfur atom. In d, the hydrogen atom is attached to a carbon atom.
So, the hydrogen atom at a position will be the most acidic hydrogen.
Therefore, the correct answer is an option (C)- a.
Note: When there is a double bond, the hydrogen atom is more acidic than the single bond because the s-character increases and the triple bond has more acidity as the s-character in the triple bond is the highest.
