Question
Question: (i) Ethanoic acid to ethanamine (ii) p-toluidine to 2-bromo-4-methylaniline. (iii) methyl bromi...
(i) Ethanoic acid to ethanamine
(ii) p-toluidine to 2-bromo-4-methylaniline.
(iii) methyl bromide to ethanamine
Solution
We can make any compound to undergo conversion into any corresponding alcohol, acid ,ether or amine only then, if we know about those compounds properly, their methods of preparation, their physical-chemical properties and their chemical reactions. So, recall the reactions of the above given compounds properly and then , you can easily make their conversion into the corresponding compounds.
Complete answer:
Considering the given conversions one by one as:
(i) Ethanoic acid to ethanamine
First of all, ethanoic acid undergoes reduction to alcohols in the [presence of lithium aluminum hydride and then alcohols so formed when made to react with the metal halides i.e. phosphorus pentachloride, forms alkyl halide when on reaction with ammonia gives the ethanamine. The overall reaction occurs as;
CH3COOHLiAlH4CH3CH2OHPCl5CH3CH2ClNH3CH3CH2NH2
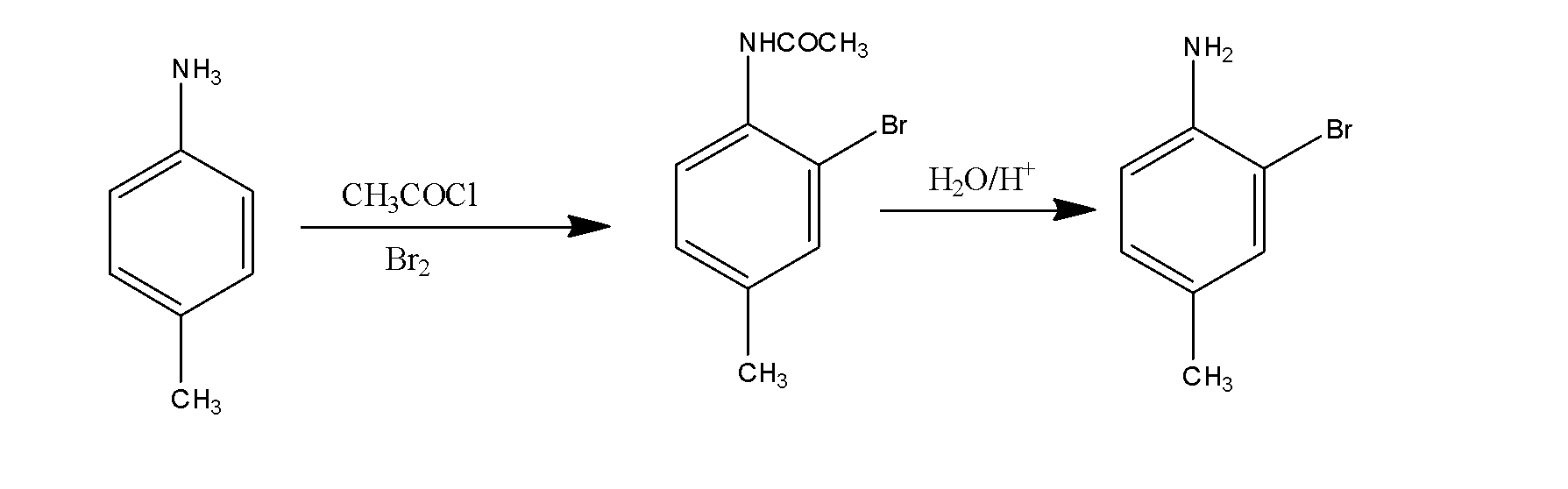
(ii) p-toluidine to 2-bromo-4-methylaniline.
In this, first of all p-toluidine reacts with the acid chloride and bromine and results in the formation of the compound which on hydrolysis results in the formation of the 2-bromo-4-methylaniline. The reaction occurs as:
(iii) methyl bromide to ethanamine
In this, first of all the methyl bromide reacts with potassium cyanide and results in the formation of the alkyl cyanide i.e. methyl cyanide and when this is made to react with the ethyl alcohol in the presence of sodium, it forms the amine i.e. the ethanamine. The overall reaction occurs as;
CH3BrKCNCH3CNNa/C2H5OHCH3CH2NH2
Note: The reaction of the alkyl cyanide with the reagent alcohol i.e. the ethanol in the presence of metal i.e. Na results in the reduction of alkyl cyanide into the primary amines and this reaction is known as the Medius reaction.
