Question
Question: How will you convert Toluene to Benzaldehyde?...
How will you convert Toluene to Benzaldehyde?
Solution
Hint: The conversion of Toluene to Benzaldehyde is used as a laboratory method in order to produce Benzaldehyde.
Complete step by step answer:
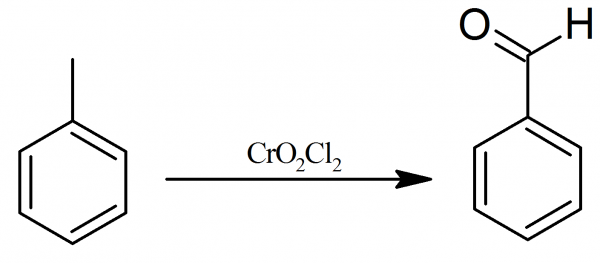
Chromyl chloride dissolved in CS2 or CCl4 is made to react with toluene in CS2 when a brown coloured product is formed. This product is decomposed with water when benzaldehyde is formed. This is Etard's reaction and is used in the laboratory for preparation of benzaldehyde.
C6H5CH3+2CrO2Cl2CCl4or CS2C6H5CH3⋅2CrO2Cl2
In case, the side-chain contains a group higher than CH3 group, then end carbon atom of the chain is oxidised by CrO2Cl2 to CHO group.
Benzaldehyde is also formed by oxidation of toluene with chromic oxide in acetic anhydride. Benzaldehyde reacts with acetic anhydride to form benzylidene diacetate which on hydrolysis with alkali or an acid yields benzaldehyde.
{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}\text{C}{{\text{H}}_{\text{3}}}\xrightarrow[{{\text{(C}{{\text{H}}_{\text{3}}}\text{CO)}}_{\text{2}}}\text{O}]{\text{Cr}{{\text{O}}_{\text{3}}}\text{, }\\!\\![\\!\\!\text{ O }\\!\\!]\\!\\!\text{ }}{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}\text{CH(OCOC}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{2}}}
Additional Information:
(i) Benzaldehyde is a colourless oily liquid. Its boiling point is 452K.
(ii) It has the smell of bitter almonds.
(iii) It is sparingly soluble in water but highly soluble in organic solvents.
(iv) It is steam volatile.
(v) It is heavier than water (sp. gr. 1.0504 at 288K)
(vi) It is poisonous in nature.
Note: Benzaldehyde is an organic compound consisting of a benzene ring with a formyl (−CHO) substituent. It is the simplest atomic aldehyde and one of the most industrially useful. Benzaldehyde is less reactive than aliphatic aldehyde.
