Question
Question: How do you draw \({C_8}{H_{18}}\) (octane)? How many isomers are there? What are the names of the is...
How do you draw C8H18 (octane)? How many isomers are there? What are the names of the isomers?
Solution
From the given molecular formula of octane C8H18, it can be known that the total number of carbon atom present in octane is 8 and the number of hydrogen atom present in octane is 18.
Complete step by step answer:
The octane is a hydrocarbon with 8 carbon atoms and 18 hydrogen atoms. The molecular formula of octane is C8H18.
The carbon is a tetravalent atom. It contains 4 electrons in the valence shell so it can donate its four electrons to form bonds with four other atoms.
The structure of octane is drawn by first drawing a straight chain or parent chain of 8 carbons and attaching 18 hydrogen on carbon to fulfill its tetravalency.
The structure of octane is shown below.
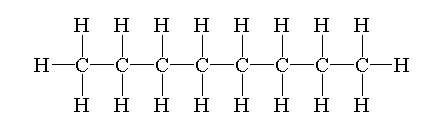
Isomers are defined as the molecules which have similar formulae but have different arrangements of atoms in the space.
Octane has a total 18 isomers.
(1) Octane
(2) 2-Methyl heptane
(3) 3-Methyl heptane
(4) 4-Methyl heptane
(5) 2, 2-Dimethyl hexane
(6) 2, 3-Dimethyl hexane
(7) 2, 4-Dimethyl hexane
(8) 2, 5-Dimethyl hexane
(9) 3, 3-Dimethyl hexane
(10) 3, 4-Dimethyl hexane
(11) 3-Ethyl hexane
(12) 2, 2, 3-Trimethylpentane
(13) 2, 2, 4-Trimethyl pentane
(14) 2, 3, 3-Trimethylpentane
(15) 2, 3, 4-Trimethyl pentane
(16) 2-Methyl-3-ethyl pentane
(17) 3-Methyl-3-ethyl pentane
(18) Tetramethylbutane
Note:
These isomers are also known as structural isomers as the structural representation of the atoms is different in each isomer but the number of atoms are same in each isomer.
