Question
Question: Draw the structure of the following compound: p,p’-dihydroxybenzophenone...
Draw the structure of the following compound:
p,p’-dihydroxybenzophenone
Solution
p,p’-dihydroxybenzophenone is an organic compound where two phenol groups are attached together with a carbonyl (-CO) group.
Complete step by step answer:
p,p’-dihydroxybenzophenone is a organic compound that have the molecular formula (HOC6H4)2CO. As it names, there are two hydroxyl groups present in the para position (fourth position) in the phenyl ring counting from the central ketone group.
It produces by the rearrangement reaction of p-hydroxyphenyl benzoate which is as follows:
HOC6H4CO2C6H5 → (HOC6H4)2CO.
Alternatively, from p-hydroxybenzoic acid, p,p’-dihydroxybenzophenone can be formed. At first, p-hydroxybenzoic acid is converted to p-acetoxybenzoyl chloride. Further the acid chloride p-acetoxybenzoyl chloride upon deacetylation reaction with phenol generates 4,4'-dihydroxybenzophenone.
The structure of p,p’-dihydroxybenzophenone is drawn and as following:
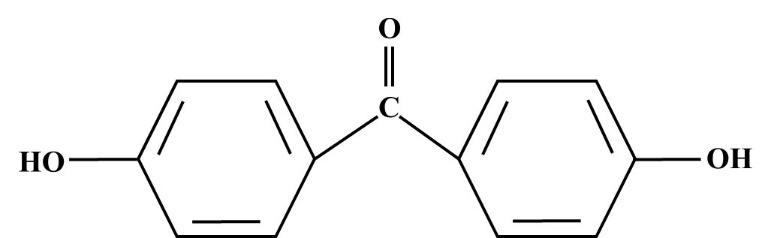
Additional information: The p,p’-dihydroxybenzophenone is a white solid product. It has many industrial application which are as follows:
-In fiber optical devices,
-In UV light stabilizers,
-In adhesives, cosmetics and plastic compounds,
-In electronic circuit boards,
-In polycarbonate polymers.
Note:
-p,p’-dihydroxybenzophenone is an organic compound that contains two phenol rings held together by a carbonyl group. The representative image is given in this solution above.
-In human physiology, the p,p’-dihydroxybenzophenone has adverse effects as it can act as an endocrine disruptor. In fungus, p,p’-dihydroxybenzophenone targets a particular enzyme Lanosterol 14-alpha demethylase. This particular enzyme catalyzes C14-demethylation of lanosterol that is an important agent for ergosterol biosynthesis. It converts lanosterol into 4,4'-dimethyl cholesta-8,14,24-triene-3-beta-ol. p,p’-dihydroxybenzophenone binds with the enzyme Lanosterol 14-alpha demethylase and blocks the lanosterol pathway. Thus by the above mechanism p,p’-dihydroxybenzophenone inhibits the fungal cell wall biosynthetic pathway and possesses anti-fungal activity.
