Question
Question: Complete the equation: 
A.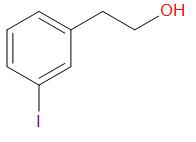
B.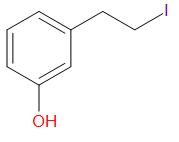
C.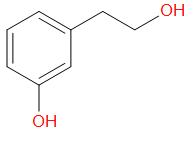
D.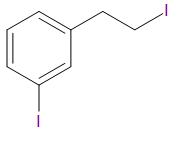
Solution
The given reaction is that for the cleavage of ethers by halogen acids. The given ether is an unsymmetrical ether. In such a case, when two different alkyl groups are present on the oxygen, then the formation of the alkyl iodide and alcohol depends on the nature of the alkyl groups attached.
Complete step by step solution:
For ease of solving and explanation, we number the two ether groups as follows.
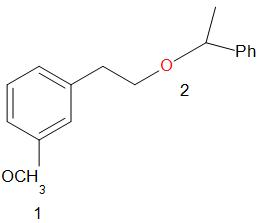
First, the oxygen atom in the ether is protonated by the acid. If both the alkyl groups attached to the oxygen are primary or secondary, the reaction proceeds by SN2 path. The iodide ion attacks the smaller of the two alkyl groups forming an alkyl iodide.
As the iodide ion attacks by the SN2 mechanism, we must consider steric factors while determining the product of the reaction. So the smaller of the two groups forms a bond with the iodide ion and the larger group forms an alcohol.
In the given compound, at the oxygen 1, the two groups are a phenyl group and a methyl group. The iodide ion will leave with the smaller of the two groups that is methyl group and an alcohol is obtained in the main product.
At the oxygen 2, of the two groups attached, one is primary while the other is a secondary group. So, the iodide ion preferably forms a bond with the primary alkyl group and a secondary alcohol leaves the molecule giving the product as,
Thus, the correct answer is B.
Note:
Ethers under vigorous conditions like concentrated acids and high temperatures undergo cleavage. The acids used are usually hydrogen bromide or hydrogen iodide. Since the reaction proceeds by the SN2 mechanism, the nucleophile, in this case halide ion, must be a strong nucleophile. Thus, we use only hydrogen iodide in the cleavage of aromatic ethers.
