Question
Question: Choose the correct option for the following: Assertion: \(NBS\) is a reagent in allylic brominatio...
Choose the correct option for the following:
Assertion: NBS is a reagent in allylic bromination.
Reason: Alkenes undergo electrophilic substitution reactions.
(A) Both assertion and reason are correct. The reason is the correct explanation for the assertion.
(B) Both assertion and reason are correct. The reason is not the correct explanation for the assertion.
(C) The assertion is correct but the reason is incorrect.
(D) Both assertion and reason are incorrect.
Solution
We know that reagent used in a chemical reaction is a compound added to the system to cause a chemical reaction. A reagent is used in allylic bromination in which a hydrogen atom that is attached to a carbon atom adjacent to a double bond is replaced.
Complete step-by-step solution: We know that reagent used in a chemical reaction is a compound added to the system to cause a chemical reaction. The reagent causes the desired chemical reaction. Here the reagent used is NBS . Its full name is N− bromosuccinimide. It is a substitute for bromine as it gives a low concentration of Br2 . It is used as a reagent in allylic bromination. Allylic bromination is the replacement of a hydrogen atom attached to a carbon atom adjacent to a double bond. The reaction occurs in presence of light. An example of this reaction is as follow:
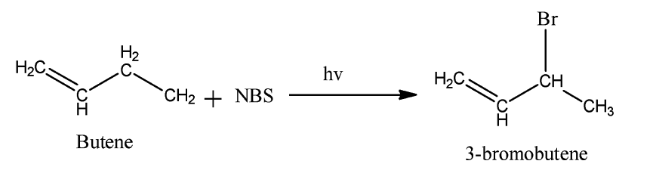
Thus, we see that the assertion is correct. Now alkenes are hydrocarbons in which there is at least one double bond between carbon-carbon atoms. Alkenes have molecular formula CnH2n . Electrophiles are electron-deficient species. Due to the presence of pi bond in alkenes, electrophiles attack alkenes and cause electrophilic addition reactions. An example of this reaction is when hydrogen bromide attacks ethane to form bromoethane.

Thus we see that the statement in reason is correct but it is not the correct explanation for the given assertion. As discussed NBS gives a low concentration of Br2 . Therefore it causes substitution reaction and prevents addition reaction.
Therefore, the correct option is B.
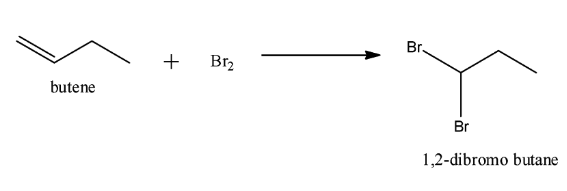
Note: It is important to know that a low concentration of bromine is needed to cause free radical halogenation reaction. We should not use Br2 directly as it will simply add to the alkene. When Br2 is added to butene instead of NBS following reaction will occur