Question
Question: Anisole on reaction with \(HI\) produces only phenol and methyl iodide. Give a reason?...
Anisole on reaction with HI produces only phenol and methyl iodide. Give a reason?
Solution
In organic chemistry, the electrophilic substitution reactions are the type of chemical reactions in which an electrophile displaces a group in a compound. This reaction involves the initial attack of an electrophile followed by the reaction with a nucleophile in order to form the final product. Most of the aromatic compounds undergo electrophilic substitution reactions due to the nucleophilic nature of the benzene ring.
Complete answer:
The reaction mechanism for the given substitution reaction is as follows:
Step-1: Dissociation of hydrogen iodide takes place into hydrogen and iodide ions respectively. The reaction is as follows:
HI→H++I−
Step-2: The lone pair present on the oxygen atom of the anisole will act as a nucleophile and an attack of hydrogen ion will take place. The reaction proceeds as follows:
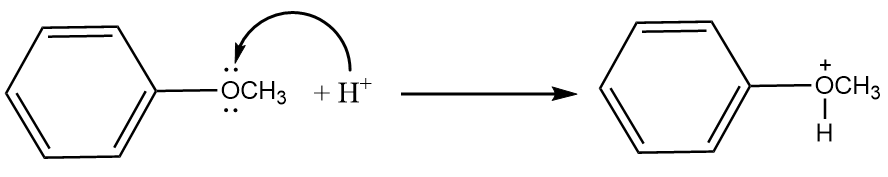
Step-3: Iodide ions will attack the methyl group and formation of phenol and methyl iodide will take place. The reaction proceeds as follows:
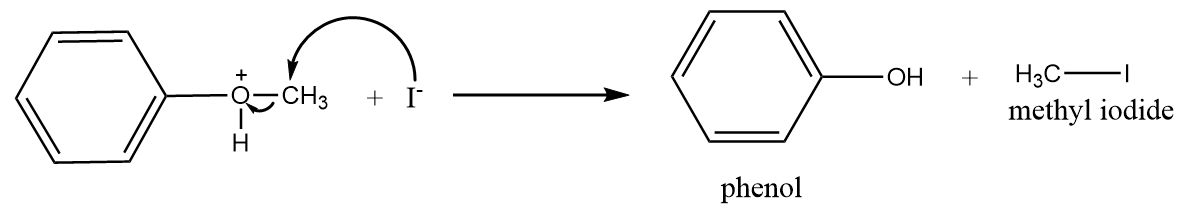
In the reaction process, only phenol and methyl iodide are formed because the bond between O−CH3 is weaker as compared to O−C6H5 due to the presence of partial double bond character. Therefore, when iodide ion attacks the cleavage of O−CH3 bonds take place to form methyl iodide instead of cleavage of O−C6H5 to form iodobenzene and methyl alcohol.
Note:
It is important to note that phenols do not further react to give halides because the carbon atoms of the ring are sp2 hybridized and consist of a cloud of pi electrons over the bonds. So, it cannot undergo a nucleophilic substitution reaction to form respective halides.
