Question
Question: A compound \({M_p}{X_q}\) has cubic-close packing (c.c.p) arrangement of X. Its unit-cell structure ...
A compound MpXq has cubic-close packing (c.c.p) arrangement of X. Its unit-cell structure is shown below. The empirical formula of the compound is:
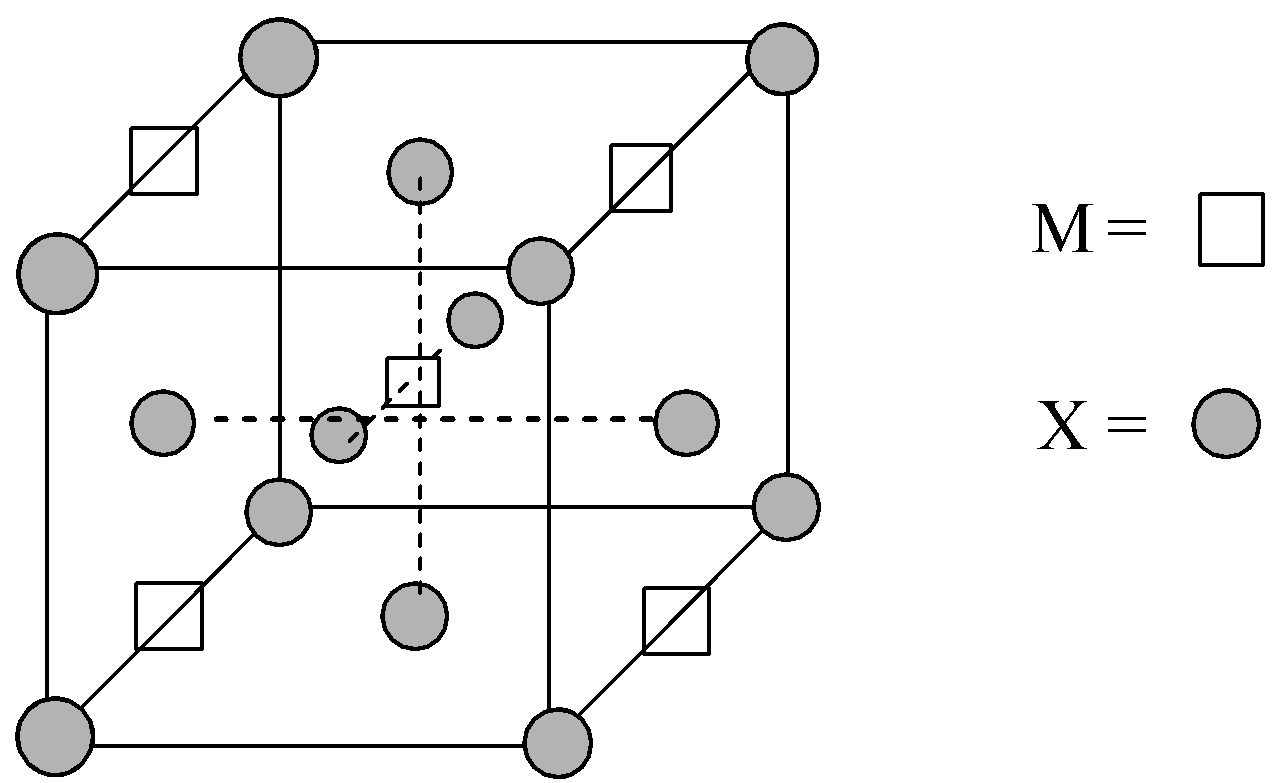
A) MX
B) MX2
C) M2X
D) M5X14
Solution
Count the number of M atoms and X atoms at different positions in the given structure of the unit-cell. To answer this question, you should know the contribution of an atom per unit cell, when the atoms are located at the corners, face-centre, and the edge-centre.
Complete step by step answer:
Given structure of the unit cell of the compound MpXq:
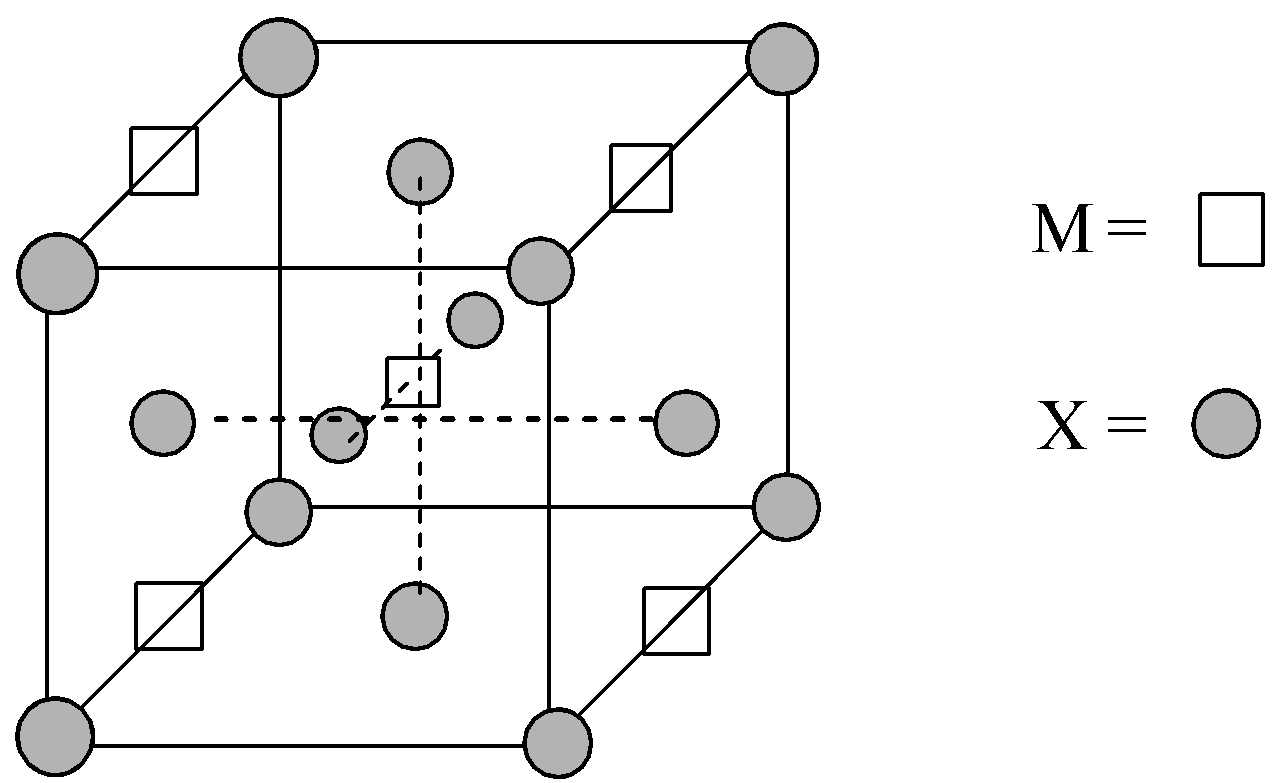
Now, let us understand the positions of X atoms:
Number of X atoms located at the corners = 8,
Number of X atoms located at the face-centres = 6
And, contribution of an atom located at the corner in a unit cell = 81 per unit cell.
Contribution of an atom located at the face-centre in a unit cell = 21 per unit cell.
Thus, total number of X atoms in the unit cell = 8 corner atoms ×81 atom per unit cell + 6 face - centred atoms × 21 atom per unit cell
∴ Total number of X atoms = 1+3=4 atoms
Now, there are four M atoms located at the edge-centres and one atom is located at the body centre of the unit cell.
Contribution of an atom located at the edge centre in a unit-cell = 41 per unit cell.
Contribution of an atom located at the body-centre in a unit-cell = 1 per unit cell.
Thus, total number of M atoms in the unit cell = 4 edge - centred atoms × 41 atom per unit cell + 1 body - centred atom × 1 atom per unit cell
∴ Total number of M atoms = 1+1=2 atoms
So, there are a total two M atoms and four X atoms in the given unit cell structure. Thus, the empirical formula of the compound is M2X4 or on simplifying, MX2.
So, the correct answer is “Option B”.
Note: Cubic close packed (ccp) unit cell or face-centred cubic (fcc) unit cell are the same unit cells but two different names. A face-centred unit cell contains atoms at all the corners and at the centre of all the faces of the cubic unit cell. A key point to note is that each atom located at the face centre is shared between two adjacent unit cells and hence only 21 of each atom belongs to the unit cell.
