Question
Question: 3-phenylpropanoic acid is the IUPAC name of which of the compound? A.Cinnamic acid B. Mendalaeic...
3-phenylpropanoic acid is the IUPAC name of which of the compound?
A.Cinnamic acid
B. Mendalaeic acid
C.Pyruvic acid
D.Citric acid
Solution
Compound with the name 3-phenylpropanoic acid contains a benzene ring and it is a crystalline compound. It is used as a flavoring agent.
Complete step by step answer:
Let us look at the molecular formula of each of the options given to us:
Cinnamic acid is an organic compound. The molecular structure of cinnamic acid is as follow:
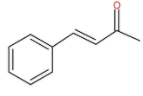
The IUPAC name of cinnamic acid is (2E)-3-Phenylprop-2-enoic acid. It also occurs in many plants naturally. It is used as an intermediate for biosynthesis and is obtained from oils of cinnamon. It is also found in Shea butter. It is also used in Indigo production and other pharmaceuticals. It is mainly used to produce methyl cinnamate, ethyl cinnamate and benzyl cinnamate for the use of perfumes. It also acts as a sweetener.
The molecular structure of Mandelic acid Is as follow:
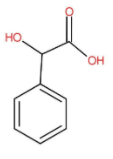
The IUPAC name of mandelic acid is hydroxyl(phenyl)acetic acid.
The molecular formula of pyruvic acid is as follow:
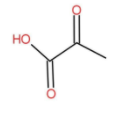
The IUPAC name is 2-Oxopropanoic acid. It is an Alpha keto acid and has no benzene ring.
The structure of citric acid is as follow:
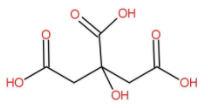
The IUPAC name is 2-hydroxypropane-1,2,3-tricarboxylic acid. Citric acid naturally occurs in various fruits and vegetables. Lemons have considerable high concentration of citric acid. It is also contained in grape food, oranges. Mainly citric acid is used as a flavoring agent and as a preservative in food and beverages specially used in soft drinks and candies.
Hence, the correct option is option A.
Note:
There are various methods of preparation of cinnamic acid. It was first prepared by base catalysed condensation with benzaldehyde and acetone chloride followed by the hydrolysis. It can also be prepared using Knovengel condensation reaction, using benzaldehyde and malonic acid along with some weak base then acid catalysed decarboxylation. It can also be prepared with the help of oxidation of cinnamaldehyde.
